
Introduction
Guarana is a plant native from the Amazon. It is best known today for the amount of caffeine it contains. The seed contains one of the highest existing levels of caffeine. This characteristic makes this species particularly interesting all over the world having first been used in beverages in traditional use. Today, we recognize in Guarana stimulating activities, in particular for it´s high caffeine content. It´s also used for concentration and memory and also for the antibacterial and fungicidal activities linked to the presence of condensed tannins and certain monomers such as catechin and epicatechin. However, as always, when the commercial use is very important and there is a significant financial stake, frauds emerge. This is the case for this species whose dry extracts from the seed are often spiked with caffeine which can be synthetic or natural.
Study
State of the art
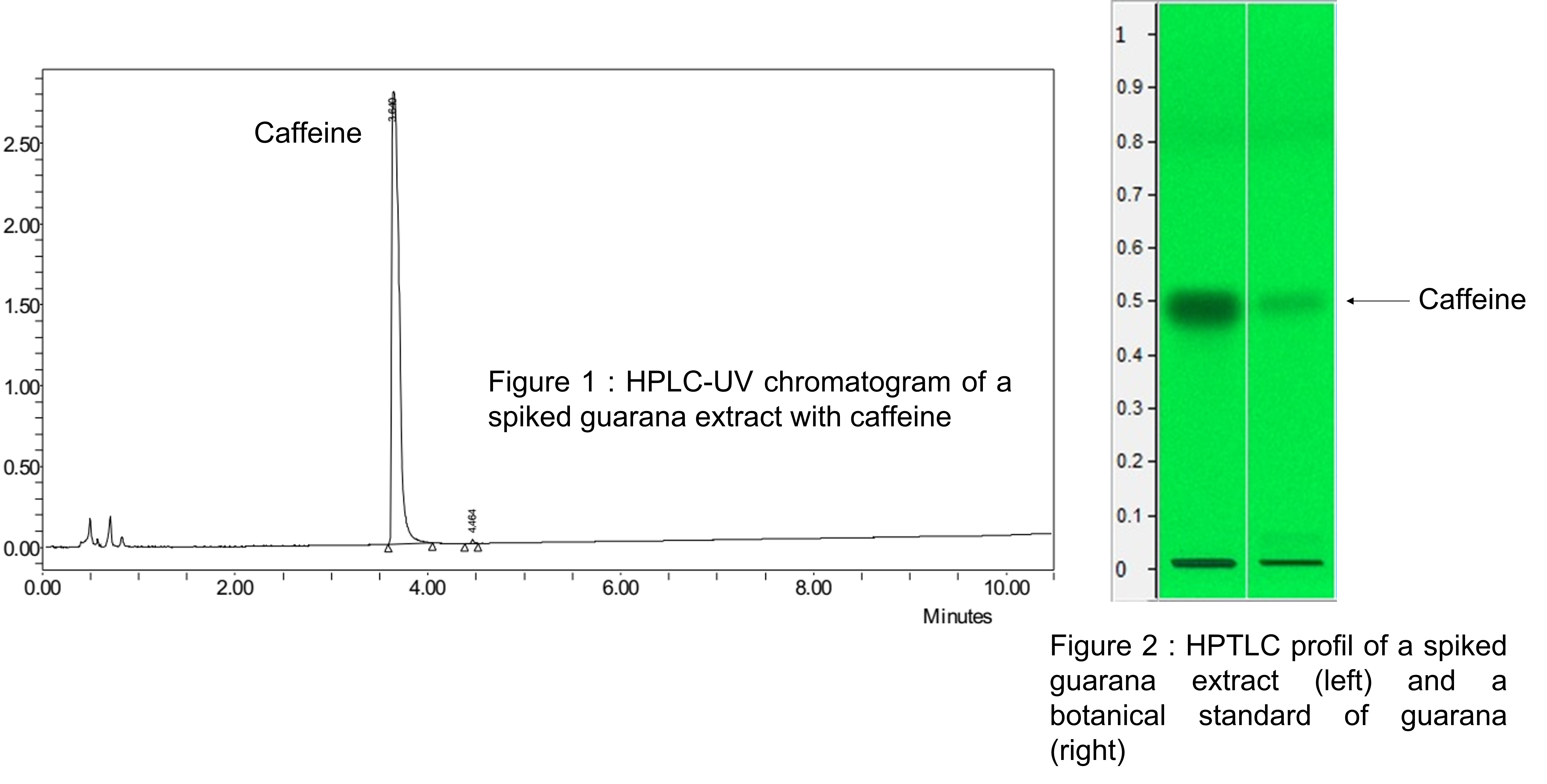
Guarana dry extracts are very often significantly enriched in caffeine which let only to detect this molecule in the final extract. (Figure 1). The Guarana seed is not only made up of caffeine. It is therefore obvious that a dry extract of Guarana enriched with caffeine will therefore not have the traditional activity that should be associates with Guarana. The benefits provided will therefore be exclusively related to the presence of caffeine. We should therefore no longer speak of dry extract of Guarana but rather of caffeine on a maltodextrin type support.In addition, to carry out controls on dry extracts, laboratories often rely on the presence of caffeine through a TLC or HPTLC analysis (Figure 2). As the analysis only focuses on the detection of caffeine, it is therefore easy to imagine that these tests will never be able to differentiate a good quality dry extract from a sample containing only caffeine on a support because the compounds other than caffeine will not be detected.
Analysis of customer samples
How can you perfom a spiked caffeine detection? A technique for this type of problem is IRMS, which is isotopic ratio mass spectrometry will measure the relative abundance of the different isotopes of a given chemical element within a sample (13C). The isotopic ratios are different for a synthetically obtained molecule and a natural molecule. The disadvantage of this technique is that the fraud of a sample which contains only synthetic caffeine can be detected, but in the case where a less significant quantity of synthetic caffeine is added, the ratios will be much more complex to interpret. And if natural caffeine has been added, the complexity is even greater.
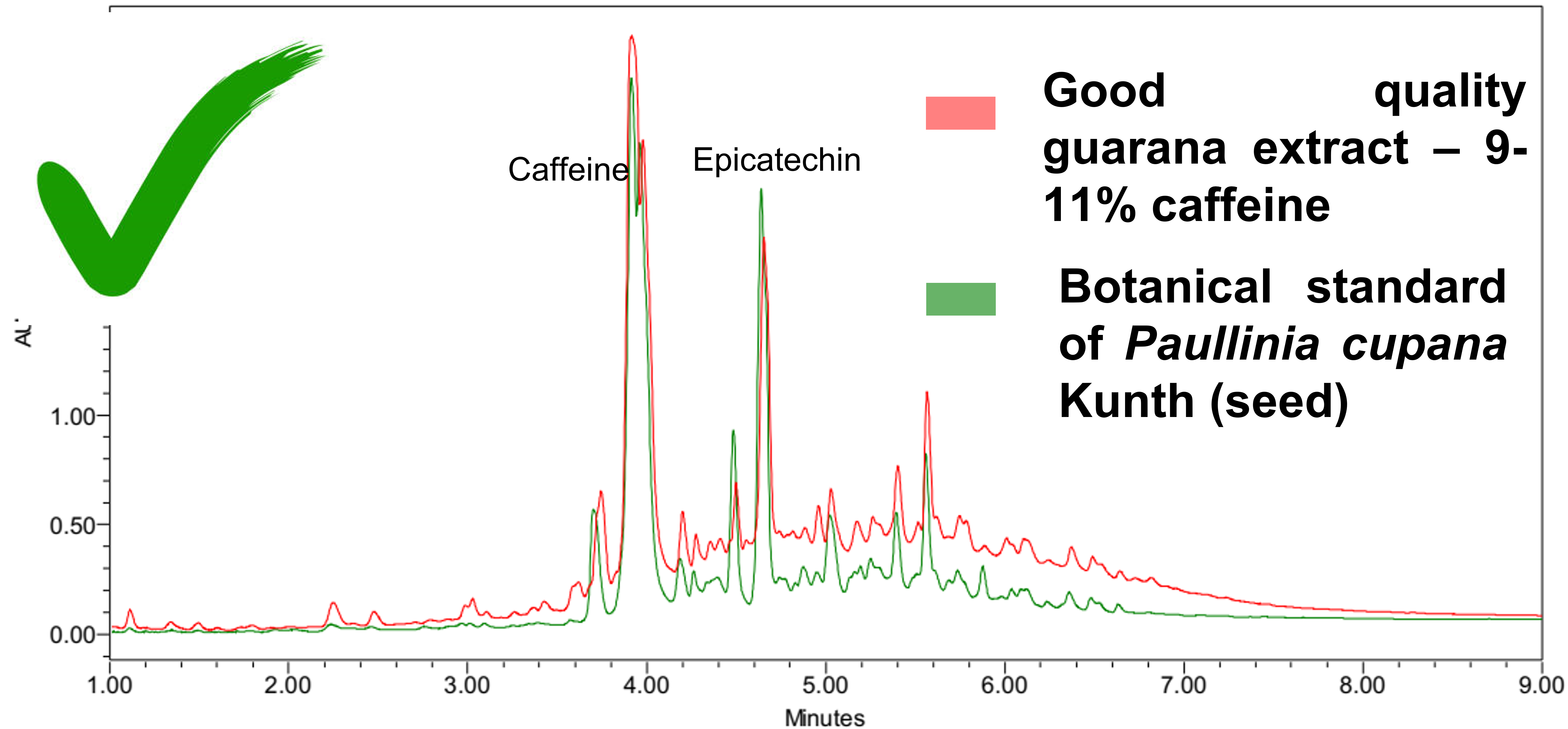
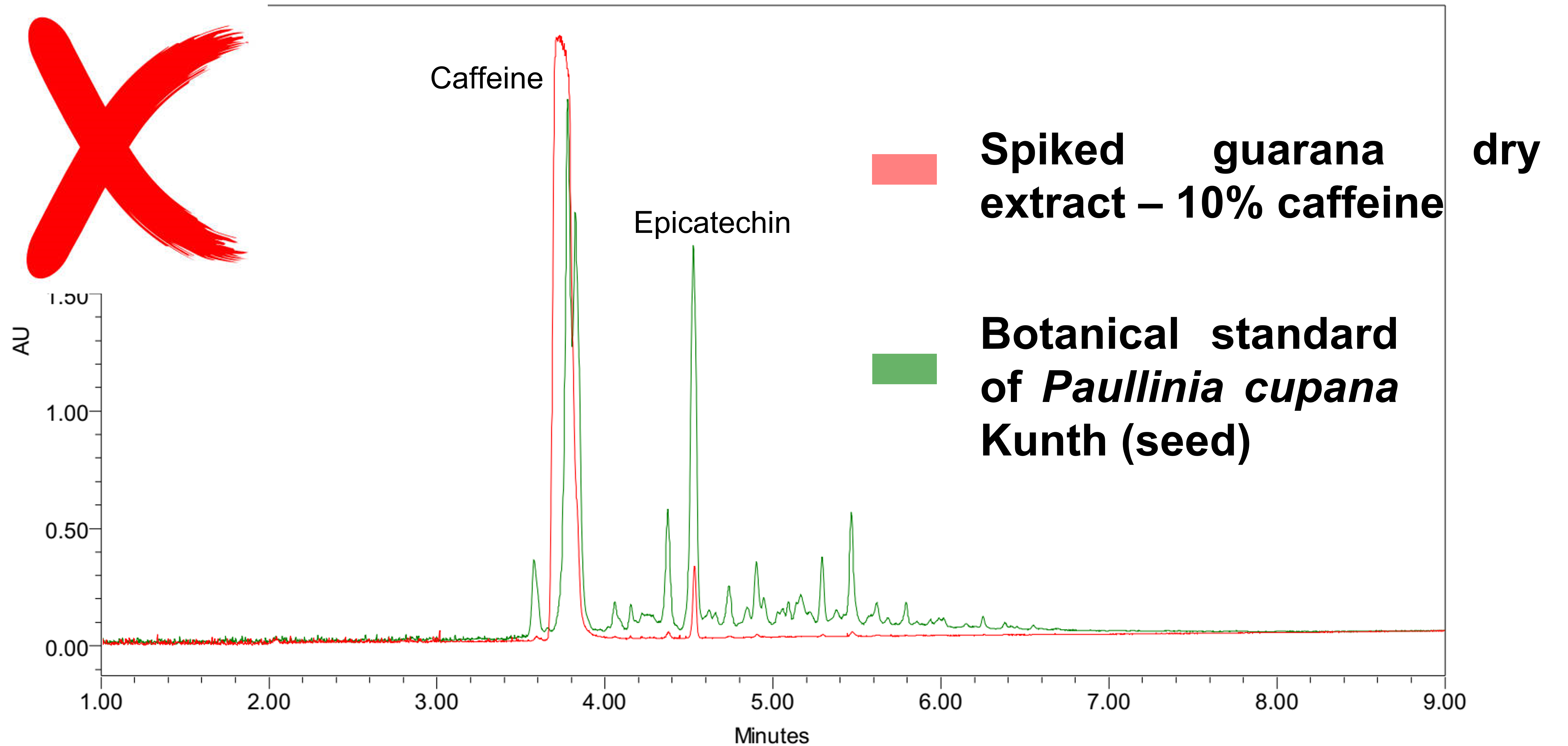
At BotaniCERT, we consider that if caffeine (natural or synthetic) has been added, the ratio between caffeine and minority compounds will change. A database is necessary to sort a caffeine enrichment out. From 58 samples analyzed during a campaign, 36 samples were identified as being formally significantly enriched in caffeine : 62% non-compliance (bottom graph). It would be easy to say that the dry extracts very concentrated in caffeine see the concentration of the minority compounds decrease in favor of the caffeine but nevertheless, many dry extracts titrated to 10% exist and present a completely expected chemical composition (top Graph) so quality samples do exist.
Conclusion
Carrying out an unsuitable assay (based only on a few criteria such as caffeine) can prove to be worse than not carrying out a control. In this particular case of the guarana, fraud is often difficult to detect and an accurate analysis should be performed.

